|
|
Post by ng on Sept 16, 2014 15:20:13 GMT -5
Awesome man!
|
|
|
|
Post by roraima on Sept 16, 2014 15:35:36 GMT -5
Great job Sokkos! I would love to get into TC if I had the space.
That VFT seedling from dvg is looking quite robust and bigger than any of my germinates. Very nice to see the D. auriculata seedlings from Shoggs as well, I'm just sowing my seeds today in hopes for an au naturel germination outside.
|
|
|
|
Post by Maiden on Sept 16, 2014 16:23:29 GMT -5
Nice ! Well done  The tiny "pretty rosette" seedlings are so cute  |
|
|
|
Post by sokkos on Nov 1, 2014 21:11:56 GMT -5
Just a little thing I noticed with my D. capensis cultures: D. capensis alba has white root tips! I multiply my D. capensis by taking side shoots, cutting off the roots and bottom leaves, then rerooting the shoots in fresh media. I usually see root growth within 10-14 days in the form of red root tips when I look under the plate. I was curious as to why my D. capensis alba didn't seem to be rooting. It wasn't until I took a closer look that I saw the root tips were in fact growing, but they just were not red. California Carnivores says this form of D. capensis is an anthocyanin mutant so its flowers are also white. It would be interesting to see if there is a difference in prey capture efficiency between the typical form and the alba form: I foresee an experiment with fruit flies coming up! As an aside, does anyone know if alba is officially a form or cultivar or some other botanical classification of D. capensis? I put "alba" in double quotations because I wasn't sure. 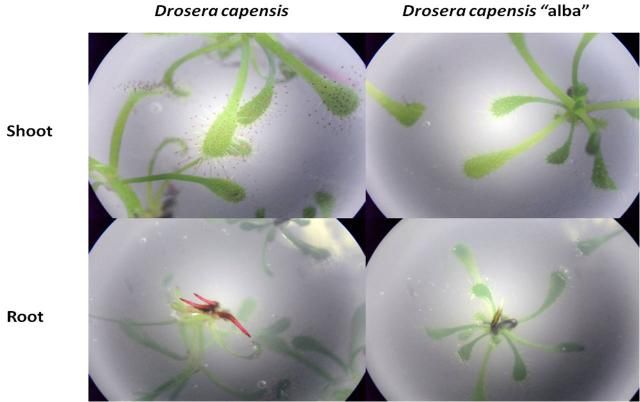 |
|
|
|
Post by canuk1w1 on Nov 2, 2014 16:24:12 GMT -5
I have some anecdotal evidence to support that theory. I have 'alba' growing adjacent to 'red' and 'red' seems to be more attractive. FWIW, mine is not the antho free variant (there is the slightest reddish hue at certain angles) but its flowers are pure white.
And of course, well done!
|
|
|
|
Post by sokkos on Dec 5, 2014 0:31:24 GMT -5
Update: December 4th Drosera binata. I can't seem to get these to grow leaves. All they seem to do is put out flower stalks and roots.  D. tokaiensis flowering  D. auriculata doing something! For the longest time all my seedlings did were grow into small compact mounds of leaves and didn't root into the media. I am trying some media variants to see if I can get these to grow for me.  D. filliformis. Now one of my favourite species to look at in culture. These grow fast!  Not a CP but this is an African violet. In one of the labs, the students were learning about somatic embryogenesis using African violet leaves. I decided to do the lab exercise with them and this is a tiny plant that has developed from a somatic embryo. The second picture is for size reference. I was planning to give these out as tiny gifts to people.   |
|
|
|
Post by ng on Dec 5, 2014 1:02:56 GMT -5
Are those those cultures from the filiformis seed I sent sok?
|
|
|
|
Post by sokkos on Dec 5, 2014 9:46:55 GMT -5
Are those those cultures from the filiformis seed I sent sok? They sure are! All the seeds that you've given me had excellent germination! |
|
|
|
Post by ng on Dec 5, 2014 11:35:37 GMT -5
Niiiiiiiiice
|
|
|
|
Post by lloyd on Dec 5, 2014 16:07:39 GMT -5
Very interesting. Are there hormones in the binata? I do see some leaves there. I like the auriculata.
|
|
|
|
Post by Dennis A(cook1973) on Dec 5, 2014 17:52:02 GMT -5
Just a little thing I noticed with my D. capensis cultures: D. capensis alba has white root tips! I multiply my D. capensis by taking side shoots, cutting off the roots and bottom leaves, then rerooting the shoots in fresh media. I usually see root growth within 10-14 days in the form of red root tips when I look under the plate. I was curious as to why my D. capensis alba didn't seem to be rooting. It wasn't until I took a closer look that I saw the root tips were in fact growing, but they just were not red. California Carnivores says this form of D. capensis is an anthocyanin mutant so its flowers are also white. It would be interesting to see if there is a difference in prey capture efficiency between the typical form and the alba form: I foresee an experiment with fruit flies coming up! As an aside, does anyone know if alba is officially a form or cultivar or some other botanical classification of D. capensis? I put "alba" in double quotations because I wasn't sure.  Hey man, if you decide to sell one get at me via pm |
|
|
|
Post by Seymour on Dec 5, 2014 21:43:57 GMT -5
This is an awesome job you've done here bud. I planned trying tc with my orchids but I never did. I don't think I have the gear to provide a sterile enough environment. Do u mix anything else with the agar?? I know with some orchid seeds there is no Starter nutrients so the agar mix contains the essentials to sustain the seed. But CP's have that contained in the seed. Does this lower the chances of fungus? Sry for the newbie questions but The process intrigues me.
JB
|
|
|
|
Post by ng on Dec 5, 2014 21:47:29 GMT -5
I'm soooo in to this.
|
|
|
|
Post by sokkos on Dec 6, 2014 0:26:27 GMT -5
Very interesting. Are there hormones in the binata? I do see some leaves there. I like the auriculata. No hormones in the binata. Just half strength MS. There are a few leaves, but as the culture gets older, the leaves keep shrinking and eventually they only put out flower stalks. I'm thinking of maybe putting some auxins in the media and try to get the plantlets to root, then transfer them out of culture before they start flowering. I've put some onto 1/4MS to see if that will make a difference. At this point it's guess work because I'm using the same media as what's reported in the literature. |
|
|
|
Post by sokkos on Dec 6, 2014 0:39:07 GMT -5
This is an awesome job you've done here bud. I planned trying tc with my orchids but I never did. I don't think I have the gear to provide a sterile enough environment. Do u mix anything else with the agar?? I know with some orchid seeds there is no Starter nutrients so the agar mix contains the essentials to sustain the seed. But CP's have that contained in the seed. Does this lower the chances of fungus? Sry for the newbie questions but The process intrigues me. JB I've done phalaenopsis green pod before. I cut the pod off, then surface sterilized the whole pod using bleach. Then I cut open the pod and scraped the seeds onto media. I was successful, but I have a feeling the pod wasn't mature enough. I also didn't use special orchid media, but was still able to get quite a few seedlings going. The media I used back then was normal MS media (4.4g/L + B5 vitamins + 8g/L agar + 30g/L sugar). The different orchid medias differ slightly, namely there is less nitrogen. You may want to look up Knudson, White, or Vacin and Went media. In hindsight it would have been best to cut down and use half strength MS instead of full strength. Some people use mashed banana or potato or coconut water in their media. You are right in that the majority of orchid seeds do not have endosperm to sustain the seed as it germinates. I would argue orchid seeds from an unopened pod is less likely to be contaminated versus cp seeds. |
|